by William J. Bailey, M.P.H.
The punch line to an often told joke suggests that the reason medical journals use technical jargon is to make sure that only doctors understand the message. With regard to recent medical journal discussions of alcoholic beverage doses and blood alcohol levels, however, a not-so-funny result of the latest extreme in technical jargon is that even doctors often don't understand the message. In an attempt to develop an "international standard" for measurements described in scientific publications, the so-called Systeme Internationale requires that journals describe doses of beverage alcohol in terms of "grams," rather than "ounces," and the blood levels are described in terms of "millimoles per liter," instead of the usual "percent of alcohol by volume" measurement.
This means that, instead of describing the ingestion of two, twelve ounce beers containing one ounce of absolute alcohol, the medical journal speaks about ingestion of 28.5 grams alcohol. Instead of describing the level of alcohol in a patient's blood as 0.10% (or 100 mg/dL) the level is described as 22 mmol/L! This system certainly is confusing for the layperson, and is likely to create comprehension problems for many physicians as well.
 A "millimole"
is one one-thousandth of a "mole." A "mole" is defined as
the amount of a substance contained in exactly 6.02 x
A "millimole"
is one one-thousandth of a "mole." A "mole" is defined as
the amount of a substance contained in exactly 6.02 x
Add to this discussion the differences between measures of the percentage of alcohol "by volume" versus "by weight," different systems of measuring the "proof" of an alcoholic beverage, medical versus legal standards for measuring blood alcohol levels, and discussions of calorie content and specific gravity. If you are confused, you are not alone.
Further complicating discussions of amounts of alcohol is the difference between potency measures based upon "percent of alcohol by volume" and measures based upon "percent of alcohol by weight." Since alcohol weighs less than water, any discussion about the proportion of alcohol in a beverage must specify whether the comparison is "by weight" or "by volume." A pint of water weights about one pound (16 ounces). A pint of pure alcohol weighs about 12.8 ounces. If one pint of pure alcohol is mixed with one pint of water, the result is a beverage that is 50% alcohol "by volume." If one pound of pure alcohol is mixed with one pound of pure water, the result is a beverage that is 50% alcohol "by weight." Since one pound of water 16 fluid ounces, and one pound of alcohol is about 20 fluid ounces, the 36 total fluid ounces in a resulting beverage is about 55.5% alcohol "by volume," although it is 50% "by weight." As an example of the importance of specifying the system used to measure the percentage of alcohol in a beverage, Coors Brewing Company promotional materials describe "Zima" as containing 3.7% alcohol by weight; 4.6% alcohol by volume. To convert measures by weight into measures by volume (or visa versa), use a conversion factor of 0.79.
The strength of an alcoholic beverage is usually expressed as either the percentage of absolute alcohol by volume, or as "proof." In the USA, proof is a measure of the amount of absolute alcohol in distilled spirits, and is calculated as the number of parts of alcohol in 200 parts of beverage at 60 degrees Fahrenheit (a beverage that is 50% alcohol is "100 proof;" a beverage that is 43% alcohol is "86 proof"). Under the American system, one "proof" is equal to one-half percent of alcohol by volume.
The concept of "proof" emerged prior to the 18th century, when few people had the equipment necessary to make precise measurements of the percentage of alcohol in a beverage. Exotic formulas, such as igniting a mixture of equal parts of the beverage and gunpowder, were used to test the proof of distilled spirits.
A different system for measuring proof is used in England, In England, proof is measured by comparing equal volumes of water and the beverage at 51 degrees Fahrenheit. 100 proof is set at the point at which the beverage weighs 12/13th as much as water. This means that a beverage that is said to be 100 proof under the British system is 114.2 proof under the American system. A beverage that is 100 proof under the American system is said to be 87.6 proof under the British system.
The dose of alcohol is calculated by multiplying the volume of an alcoholic beverage by the percentage of alcohol by volume. For example, twelve ounces of beer that is 4% alcohol by volume would have a dose of 0.48 ounces of alcohol. Pure alcohol (100% alcohol) is called "absolute alcohol." Absolute alcohol is alcohol without any water molecules in it. Alcohol produced by distillation contains water molecules that are very hard to remove. The strongest readily available alcoholic beverage, often called "neutral grain spirits" is about 95% absolute alcohol (190 proof). It is possible to manufacture absolute alcohol, also called "scientific alcohol," by synthesis.
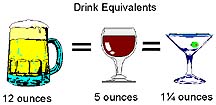
The dose of alcohol in a typical 12 ounce can of beer, is approximately equal to the dose of alcohol in a 4 to 5 ounce serving of wine, or in a "shot" of whiskey. This average-sized dose of alcohol is equal to one-half ounce of absolute alcohol, and is sometimes called a "drink equivalent."
| Examples of Drink Equivalence | |
|---|---|
| 12 ounces of 4% beer | 0.48 ounces of absolute alcohol |
| 5 ounces of 10% wine | 0.50 ounces of absolute alcohol |
| 1.25 ounces of 40% vodka (80 proof) | 0.50 ounces of absolute alcohol |
| 1.25 ounces of 43% whiskey (86 proof) | 0.52 ounces of alsolute alcohol |
| all of the above servings have approximately 0.50 ounces of absolute alcohol | |
Some typical servings of other alcoholic beverages do NOT contain a single drink equivalent. They may contain more or less alcohol than is found in an "average-sized" dose of alcohol. Examples include:
| Examples of Drink Non-Equivalence | ||
|---|---|---|
| Dose of Alcoholic Beverage | Amount of absolute alcohol | Number of drink equivalents |
| 40 ounce bottle of 8% malt liquor | 3.2 oz. | 6.4 drink equivalents |
| 12 ounce bottle of 2.5% low alcohol beer | 0.3 oz. | 0.6 drink equivalents |
| 1.25 ounce shot of 151 proof rum | 0.94 oz. | 1.9 drink equivalents |
| 12.5 ounce bottle of 20% fortified wine cooler | 2.5 oz. | 5.0 drink equivalents |
One fluid ounce (U.S.) of alcohol equals:
Common measurements of alcoholic beverages(U.S.):
| Common Measurements for Alcoholic Beverages | |
|---|---|
| "pony shot" | 0.5 jigger; 0.75 fluid ounces |
| "shot" | 0.666 jigger; 1 fluid ounce |
| "large shot" | 1.25 ounces |
| "jigger" | 1.5 shots; 1.5 fluid ounces |
| pint | 16 shots; 0.625 fifths |
| fifth | 25.6 shots; 25.6 ounces; 1.6 pints; 0.8 quarts; 0.75706 liters |
| quart | 32 shots; 32 ounces; 1.25 fifths |
| magnum | 2 quarts; 2.49797 wine bottles |
| bottle wine | 0.800633 quarts; 0.7577 liters |
One gram of ethyl alcohol yields 7 calories of energy when metabolized by the body. That converts of approximately 200 calories per ounce of absolute alcohol, or about 100 calories per drink equivalent (one-half ounce of absolute alcohol). The total calorie content of an alcoholic beverage includes the calories from the alcohol itself, and calories from other components of the beverage (residual sugars or grains in beers and wines, for example). A typical serving of regular beer yields about 150 calories (about 100 calories from the alcohol and about 50 calories from the residual carbohydrates). A typical serving of light beer has a lower alcohol content and less residual carbohydrates, so the total calorie content could be about 100 calories (about 80 calories from the alcohol and about 20 calories from the residual carbohydrates).
The most common system for measuring and reporting Blood Alcohol Levels (BAL) is calculated using the weight of alcohol (milligrams) and the volume of blood (deciliter). This yields a Blood Alcohol Concentration that can be expressed as a percentage (ie. 0.10% alcohol by volume), or as a proportion (ie. 100 mg. per deciliter). This system is the one prescribed by almost every state, and is sometimes referred to as the "weight by volume" or "w/v" method.
A few states prescribe a "weight by weight" or "w/w" method (milligrams of alcohol in milligrams of blood). For forensic evidence purposes, the w/w can be calculated by dividing the w/v by a factor or 1.055 (the average specific gravity of blood).
Although most evidentiary rules specify that the proportion of alcohol in the whole blood be used to measure the level of intoxication, most hospitals routinely calculate the proportion of a drug in the plasma portion of the blood. While this measurement is the most useful one for medical purposes, it is not the appropriate one for most legal purposes. Since the addition of red and white blood cells would add to the blood volume that dilutes the alcohol, using only the plasma fraction would result in a percentage of alcohol that is higher than would be found in whole blood. Although there is some variation from person to person, depending upon the density of their bloods, the average adjustment factor is 1.16. In other words, to estimate the alcohol level in whole blood using the alcohol level in blood plasma, divide by 1.16. For example, a plasma alcohol level of 0.15% would convert to a blood alcohol level of 0.13%.
| Comparison of Blood Alcohol Level Reporting Systems | |
|---|---|
| Six different methods of reporting the same blood alcohol measurement. | |
| Method of Stating the Measurement | Result |
| Blood Alcohol Concentration (percent weight to volume) | 0.10% |
| Milligrams per milliliter | 1.0 |
| Milligram percent (milligrams per 100 ml.) | 100 |
| parts per million | 1000 |
| Percent by weight (weight by weight) | 0.09% |
Alcohol and the Impaired Driver: A Manual on the Medicolegal Aspects of Chemical Tests for Intoxication with Supplement on Breath/Alcohol Tests. Chicago: National Safety Council, 1976.
American Medical Association. Manual of Style, 8th ed. Baltimore: Williams and Wilkins, 1989.
Bailey WJ. Drug Use in American Society, 3rd ed. Minneapolis: Burgess Publishing Group, 1993.
DeLeo JD. Fundamentals of Chemistry: General, Organic, and Biological. Glenview, IL: Scott, Foresman and Company. 1988.
Gerald MC. Pharmacology: An Introduction to Drugs, 2nd ed. Englewood Cliffs, NJ: Prentice-Hall, 1981.
Gilman AG et al., Goodman and Gilman's The Pharmacological Basis of Therapeutics, 9th ed. New York: Macmillan Publishing Company, 1995.
O'Brien R, Chafetz M. The Encyclopedia of Alcoholism. New York: Facts-on-File, 1982.
Indiana Prevention Resource Center
Factline on Alcohol Doses, Measurements, and Blood
Alcohol Levels
Factline Number 11, November, 1995
Indiana Prevention Resource Center
at Indiana University
840 State Road 46 Bypass, Room 110
Bloomington, IN 47405
(812) 855-1237
The opinions expressed herein are those of the author and are not necessarily those of the Trustees of Indiana University or of the Indiana Family and Social Services Administration - Division of Mental Health.